
Attorney Explains APGAR Scores for Birth Injury Evaluation
The Apgar score is a rapid assessment of your baby’s health following birth to determine if intervention is needed. Although not used to diagnose birth

The Apgar score is a rapid assessment of your baby’s health following birth to determine if intervention is needed. Although not used to diagnose birth
Bringing a child into the world should be a joyous occasion, yet for many expectant mothers, especially women of color, the experience is marred by
When signs of fetal distress are present during pregnancy or childbirth, swift action is necessary to prevent serious harm, including brain damage and death. The
Hypoxic-ischemic encephalopathy, or HIE, is a devastating medical condition affecting newborns after a traumatic birth. It occurs when the infant’s brain is deprived of oxygen

Hypoxic-ischemic encephalopathy, or HIE, is a type of birth asphyxia that occurs when a baby is deprived of oxygen before, during, or after birth. This results
During pregnancy, the umbilical cord attaches on or around the center of the placenta. This vital organ helps provide oxygen and nutrients to a growing

Premature Babies, Brain Injuries and Cerebral Palsy Birth injuries can uncover a medical mistake that was likely the cause of a preventable traumatic experience to

12 Quick Facts That Could Support Your Birth Injury Lawsuit According to the American Academy of Pediatrics, most birth injuries are avoidable when risks have

Spinal Cord Injuries Require Coordinated Diagnostic Infant Care Spinal cord injuries in newborns are usually due to excessive traction or rotation of the spinal cord

Medical Professionals Must Provide Sensitive Care to Avoid Infant Bone Fractures Obstetric staff who choose to use excessive force during labor and delivery may sometimes

Is Premature Infant Death Preventable? As many as 1 million deaths are the cause of preterm birth complications each year, making it the leading cause of death among

Black Infant Mortality Rate Has Remained Unchanged Since 2008 Each year in Illinois, approximately 145,000 live-born infants, and 950 infants die before their first birthday.

Birth Injuries Can Cause Shoulder, Arm, and Hand Paralysis in Newborn The brachial plexus is a bundle of nerves located in an infant’s body’s upper

Brain Injury Awareness Month Serves as a Reminder for Medical Professionals to Work Harder in Preventing Birth Injuries March is Brain Injury Awareness Month. The

The Connection Between Labor Inducing Drugs and Birth Injuries According to research provided through the National Partnership for Women & Families (NPWF), up to 50%

New Legislation Aimed to Save Lives and Reduce Disparities Impacting Mortality in Black Mothers and Black Babies Women of color, more so non-Hispanic Black mothers,

New Research May Provide Answers to Lessening Mortality Rate in Black Babies An associate professor and reproductive health equity researcher from the University of Minnesota

Childbirth Injuries Related to Unnecessary Episiotomy Episiotomies can create lasting injuries and make sexual intercourse painful, if not impossible, and prevent women from wanting to

8 Birth Injury Facts Parents Must Know According to the American Academy of Pediatrics, most birth injuries are avoidable when appropriate care is available. And

Latest CDC Data Shows U.S. Maternal Mortality Rate Highest Among Comparable Countries A recent analysis of the latest issue brief from the Centers for Disease

Do These 3 Things If You or Your Child Were Injured During Pregnancy or the Labor and Delivery Process Nearly 28,000 babies are born with a birth

6 Birth Injuries to Baby That Can Happen Due to Delayed C-Section Healthcare professionals are trained to detect when an emergency C-section is needed. Still,
Premature Babies Have Significant Birth Injury Risk of the Brain Premature babies, especially those born very early, often have complicated medical problems and face many

Cerebral palsy can be the result of a birth injury or abnormal development during or after pregnancy, caused by infection, fetal or pediatric stroke, undiagnosed

Understanding Infant Wrongful Death Even medical professionals can cause tragic labor and delivery mistakes that result in life-long injuries, stillborn, or a circumstance so dire

Illinois Boy Recovers $8 Million in Damages for Birth Injury When a baby’s delivery is delayed or the infant becomes lodged at birth, serious complications
Injuries to Infant Can Occur During Labor Induction Mistakes Many mothers are needlessly dying during labor or their babies sustaining life-altering injuries because of medical

Recognizing cerebral palsy symptoms in your baby provides the opportunity for valuable early intervention. Although there is no cure for cerebral palsy, early treatment may
Mistakes in Fetal Ultrasound Readings Can Have Devastating Effects During Labor & Delivery Most women will undergo several minimally invasive fetal ultrasounds during her pregnancy
Spinal cord injuries in newborns are usually a result of excessive traction or rotation of the spinal cord and twisting of a baby’s trunk during

A birth injury is defined as the structural destruction or functional deterioration of an infant’s body due to a traumatic event at birth. According to the

What is Congenital Syphilis? Syphilis is a chronic bacterial infection spread through sexual contact and sexual intercourse, making it a sexually transmitted disease (STD). Congenital

Obstetric violence has become yet another vexing problem added to the traumatic experiences that too many pregnant women in America face during both the prenatal
Around 450,000 babies are born preterm in the U.S. every year, according to the Children’s National Research Institute. Premature babies are often deprived of oxygen caused

New Illinois Law Says Every Woman Has Safe Pregnancy and Childbirth Rights New data published on January 30, 2020, by the Centers for Disease Control

Fort Campbell Hospital Birth Injury Ends With $15.1M Settlement On January 10, 2005, Kelly D. Wilson gave birth to her son at Blanchfield Army Community

Mom Dies After Routine Caesarean Section, and Dad Sues Cedars-Sinai Cedars-Sinai Medical Center in Los Angeles is facing national backlash after a woman internally bled

Understanding Infant Injuries Related to Dangerous Breech Deliveries A breech birth occurs when an infant enters the birth canal in a manner other than head

When Mothers Are Mistreated Because of Race, Babies Are at Risk of Preterm Birth and Harmful Injuries According to the U.S. Centers for Disease Control

15 Things Parents May Not Know About Their Baby’s Birth Injury A birth injury is defined as the structural destruction or functional deterioration of an

The routine induction of labor among women with preexisting type 1 and type 2 diabetes before pregnancy is a common practice and rightful under individual

In the U.S., about 1 in 33 babies is born with a birth defect each year, according to the March of Dimes. This makes birth

3 Types of Asphyxia Related Childbirth Injuries Some of the most traumatic injuries that arise during childbirth are those that affect the baby’s brain. Asphyxia

How Long Do I Have to File a Birth Injury Lawsuit in Illinois? Minor injuries to newborns may occur during the birthing process, which could

Record-Setting Verdict Against West Suburban Medical Center in Oak Park for Failure to Order Emergency Cesarean In November, an Illinois jury made up of six
4 Newborn Injury Complications Caused by Shoulder Dystocia One of the most feared complications of a normal vaginal delivery, shoulder dystocia is a frightening reality

In November, a Brooklyn, New York jury awarded 7 year old Aleigha Buck and her mother $26 million to compensate for a hospital’s failure to appropriately stall

The birth of a child is one of the happiest days in the lives of parents. However, for some, the joy can be replaced by

A Georgia jury recently awarded a couple more than $4 million in damages over the death of their infant son during a premature birthing injury. With

An Oregon family recently filed a $40 million lawsuit, stemming from injuries they allege were caused by medical malpractice on the part of doctors and hospital staff
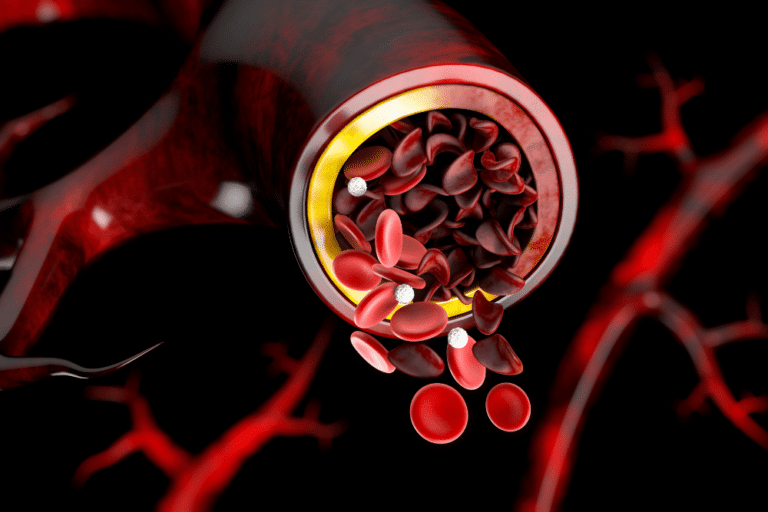
Sickle cell anemia is a form of disease where the red blood cells become hard and sticky, dying early. This causes a continuous red blood

According to a report by Medstak, a couple recently filed a lawsuit alleging medical negligence that resulted in the death of their unborn child. The complaint
The maternal forces defense is a common defense used by doctors and hospitals in medical malpractice lawsuits involving Erb’s Palsy. According to the maternal forces
The variety of medical malpractice cases is more extensive than most imagine. Considering the total number of births that occur each year, many somewhat unique

Each Chicago medical malpractice attorney at our firm works with families who have suffered brain injuries as a result of negligent medical treatment. Unfortunately, one

A recent article on MarketWatch reported that the FDA has granted approval for landmark clinical trials to study cord blood stem cell treatment for pediatric brain injuries

Over ten years ago a family’s life was forever altered following a birth injury that occurred at the Advocate Lutheran General Hospital in Park Ridge,

Levin and Perconti recently announced in a press release on November 24, 2010 that its Chicago brain attorneys reached a $6.5 million settlement for a

A jury has awarded $20.5 million in a medical malpractice suit to the family of a child with lasting medical problems caused by mistakes made

A Wisconsin jury awarded $11.4 million to the parents of a child born with Cerebral Palsy, and found a nurse and midwife at Gunderson Lutheran Medical
RUSHVILLE, IL – The community of Rushville, Illinois, is devastated after a collision involving a school bus and a semi-truck killed five people, including three
Nursing home regulations aim to guarantee that residential facilities provide safe, sanitary, home-like environments where residents receive quality care. Inadequate regulation in nursing homes has
The incident at the Judge Fisher Apartments remind us all of the vulnerabilities our cherished seniors face, even in places meant to ensure their safety.



Copyright © 2023, Levin & Perconti